Afewerki S, Wang X, Ruiz-Esparza GU, Tai C, Kong X, Zhou S, Welch K, Huang P, Bengtsson R, Xu C, and Strømme M. 12/11/2020. “
Combined Catalysis for Engineering Bioinspired, Lignin-Based, Long-Lasting, Adhesive, Self-Mending, Antimicrobial Hydrogels.” ACS Nano.
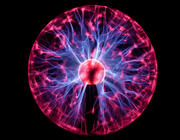
LinkAbstractThe engineering of multifunctional biomaterials using a facile sustainable methodology that follows the principles of green chemistry is still largely unexplored but would be very beneficial to the world. Here, the employment of catalytic reactions in combination with biomass-derived starting materials in the design of biomaterials would promote the development of eco-friendly technologies and sustainable materials. Herein, we disclose the combination of two catalytic cycles (combined catalysis) comprising oxidative decarboxylation and quinone-catechol redox catalysis for engineering lignin-based multifunctional antimicrobial hydrogels. The bioinspired design mimics the catechol chemistry employed by marine mussels in nature. The resultant multifunctional sustainable hydrogels (1) are robust and elastic, (2) have strong antimicrobial activity, (3) are adhesive to skin tissue and various other surfaces, and (4) are able to self-mend. A systematic characterization was carried out to fully elucidate and understand the facile and efficient catalytic strategy and the subsequent multifunctional materials. Electron paramagnetic resonance analysis confirmed the long-lasting quinone-catechol redox environment within the hydrogel system. Initial in vitrobiocompatibility studies demonstrated the low toxicity of the hydrogels. This proof-of-concept strategy could be developed into an important technological platform for the eco-friendly, bioinspired design of other multifunctional hydrogels and their use in various biomedical and flexible electronic applications.
 afewerki_et_al_acs_nano.pdf
afewerki_et_al_acs_nano.pdf Adans-Dester C, Ruiz-Esparza GU, Williamson L, and Bonato P. 8/13/2020. “
Can mHealth Technology Help Mitigate the Effects of the COVID-19 Pandemic?” IEEE Open Journal of Engineering in Medicine and Biology.
LinkAbstractGoal: The aim of the study herein reported was to review mobile health (mHealth) technologies and explore their use to monitor and mitigate the effects of the COVID-19 pandemic. Methods: A Task Force was assembled by recruiting individuals with expertise in electronic Patient-Reported Outcomes (ePRO), wearable sensors, and digital contact tracing technologies. Its members collected and discussed available information and summarized it in a series of reports. Results: The Task Force identified technologies that could be deployed in response to the COVID-19 pandemic and would likely be suitable for future pandemics. Criteria for their evaluation were agreed upon and applied to these systems. Conclusions: mHealth technologies are viable options to monitor COVID-19 patients and be used to predict symptom escalation for earlier intervention. These technologies could also be utilized to monitor individuals who are presumed non-infected and enable prediction of exposure to SARS-CoV-2, thus facilitating the prioritization of diagnostic testing.
 adans-dester_et_al_ieee_open_journal_of_engineering_in_medicine_and_biology.pdf
adans-dester_et_al_ieee_open_journal_of_engineering_in_medicine_and_biology.pdf Afewerki S, Ruiz-Esparza GU, and Lobo AO. 5/2020. “
Antimicrobial Electrospun Materials.” In Electrospun Materials and Their Allied Applications, Pp. 243-264. John Wiley & Sons.
LinkAbstractThe fast-growing public health awareness and concern of the devastating problems with bacterial infections and the mounting resistance of bacteria to conventional antibiotic treatments have made this theme the top concern. At the same time the problem will not be solved through solely inventions of antimicrobial materials preventing the prevalence of bacteria resistance. Nevertheless, the fabrication and design of these materials are highly important to find its translational applications in our daily life. In this context, electrospun materials with their inimitable advantages and facile production make them a suitable candidate for various applications. The electrospinning technology represents a versatile and facile approach for the construction of ultrathin electrospun fibers from various materials. Then, it allows the fabrication of electrospun fibers with various and controlled dimensions such as nanosized fibers which have gained significant attention due to their valuable properties such as high surface area, large porosity, and lightweight. Through the combined electrospinning and antimicrobial material employment, a very powerful, robust, and vital strategy for engineered material can be generated. These materials can be employed in many areas such as healthcare (eg, tissue repair, drug delivery, and wound healing), environmental application (eg, filters and membranes), energy applications (solar and fuels cells), and in protecting clothing for medical and chemical workers.
 afewerki_et_al_electrospun_materials_and_their_allied_applications.pdf
afewerki_et_al_electrospun_materials_and_their_allied_applications.pdf Morais AIS, Wang X, Vieira EG, Viana BC, Silva-Filho EC, Osajima JA, Afewerki S, Corat MAF, Silva HS, Marciano FR, Ruiz-Esparza GU, Stocco TD, Paula de MMM, and Lobo AO. 2/19/2020. “
Electrospraying Oxygen-Generating Microparticles for Tissue Engineering Applications.” International Journal of Nanomedicine, 2020, 15, Pp. 1173-1186.
LinkAbstractBackground: The facile preparation of oxygen-generating microparticles (M) consisting of Polycaprolactone (PCL), Pluronic F-127, and calcium peroxide (CPO) (PCL-F-CPO-M) fabricated through an electrospraying process is disclosed. The biological study confirmed the positive impact from the oxygen-generating microparticles on the cell growth with high viability. The presented technology could work as a prominent tool for various tissue engineering and biomedical applications.
Methods: The oxygen-generated microparticles fabricated through electrospraying processes were thoroughly characterization through various methods such as X-ray diffraction (XRD), Fourier Transform Infrared Spectroscopy (FTIR) analysis, and scanning electron microscopy (SEM)/SEM-Energy Dispersive Spectroscopy (EDS) analysis.
Results: The analyses confirmed the presence of the various components and the porous structure of the microparticles. Spherical shape with spongy characteristic microparticles were obtained with negative charge surface (ζ = – 16.9) and a size of 17.00 ± 0.34 μm. Furthermore, the biological study performed on rat chondrocytes demonstrated good cell viability and the positive impact of increasing the amount of CPO in the PCL-F-CPO-M.
Conclusion: This technological platform could work as an important tool for tissue engineering due to the ability of the microparticles to release oxygen in a sustained manner for up to 7 days with high cell viability.
 morais_et_al_international_journal_of_nanomedicine.pdf
morais_et_al_international_journal_of_nanomedicine.pdf Afewerki S, N Bassous, S Harb, Palo-Nieto C, Ruiz-Esparza GU, Marciano FR, Webster TJ, and Lobo AO. 1/2020. “
Advances in Antimicrobial and Osteoinductive Biomaterials.” In Racing for the Surface: Antimicrobial and Interface Tissue Engineering, Pp. 3-34. Springer.
LinkAbstractThe enormous growing problem with antibiotic resistance in pathogenic microbes is one of the greatest threats we are facing today. In the context of orthopedic applications, infections also lead to the limited healing ability of infected and defected bone. Generally, these problems are treated with a load of antibiotics or surgical intervention. Therefore, having antibacterial properties integrated with a biomaterial would reduce the time of healing and treatment, amount of antibiotic needed, and total cost. Currently, there exists several strategies and materials with the potential of tackling these challenges. Some materials with antibacterial properties currently employed are silver nanoparticles (AgNPs), cerium oxide nanoparticles (CeO2NPs), selenium nanoparticles (SeNPs), copper nanoparticles (CuNPs), antimicrobial peptides (AMPs), biopolymers (such as chitosan), and carbon nanostructures. On the other hand, osteoinductive and osteoconductive materials are important to promote bone healing and regeneration. Within this framework, materials which have been employed widely are bioactive glasses (BG), calcium phosphates (CaPs) (e.g., hydroxyapatite (HA), tricalcium β-phosphate (β-TCP), and biphasic calcium phosphate (BCP)), peptides, growth factors, and other elements (e.g., magnesium (Mg), zinc (Zn), strontium (Sr), silicon (Si), selenium (Se), and Cu, to name a few). Some of the current technological solutions that have been employed are, for instance, the use of a co-delivery system, where both the antibacterial and the osteoinducing agents are delivered from the same delivery system. However, this approach requires overcoming challenges with local delivery in a sustained and prolonged way, thus avoiding tissue toxicity. To address these challenges and promote novel biomaterials with dual action, sophisticated thinking and approaches have to be employed. For this, it is of the utmost importance to have a solid fundamental understanding of current technologies, bacteria behavior and response to treatments, and also a correlation between the material of use, the host tissue and bacteria. We hope by highlighting these aspects, we will promote the invention of the next generation of smart biomaterials with dual action ability to both inhibit infection and promote tissue growth.
 afewerki_et_al_racing_for_the_surface_antimicrobial_and_interface_tissue_engineering.pdf
afewerki_et_al_racing_for_the_surface_antimicrobial_and_interface_tissue_engineering.pdf